Hepatitis B
The hepatitis B virus is one of the most prevalent blood-borne viruses worldwide and is a major cause of chronic liver disease and liver cancer

Key messages
-
Hepatitis B is a viral infection of the liver spread through contact with blood or body fluids contaminated with hepatitis B virus.
-
Babies with the virus at birth or in their first year of life are more likely to develop persistent hepatitis B infection and chronic (long-term) liver disease.
-
The risk of contracting hepatitis B infection is low for most travellers. The risk can be reduced by avoiding unprotected sex with new partners abroad and avoiding sharing drug injection equipment, not sharing shaving equipment, avoiding acupuncture, skin piercing and tattoos (unless sterile equipment is used).
-
Hepatitis B vaccination is recommended for all travellers considered to be at risk and can be given to adults and children from birth.
-
Those who need to seek urgent medical or dental care in resource-limited countries should consider the risks from contaminated equipment or blood/blood products.
-
A sterile medical equipment kit may also be helpful when travelling to some countries.
Overview
Hepatitis B virus (HBV) is one of the most common blood-borne viruses worldwide; it can cause both an acute and chronic (long-term) infection. HBV is a major cause of liver disease and hepatocellular carcinoma (primary liver cancer), and it is estimated that worldwide 254 million people were living with chronic hepatitis B infection in 2022 [1]. Approximately 1.1 million people died due to the consequences of hepatitis B infection in 2022 [1].
Risk areas
Hepatitis B is a global health problem. According to the World Health Organization (WHO), the burden of infection is highest in the WHO Western Pacific and African Regions where 97 million and 65 million people, respectively, have a chronic HBV infection [1]. A 2012 WHO map (see below) shows many other areas of the world with an estimated intermediate to high prevalence of chronic hepatitis B infection in the local population. For country specific information and vaccine recommendations please see our Country Information pages.
Map of countries or areas with intermediate to high prevalence (> 2% of the population) of hepatitis B infection, 2012
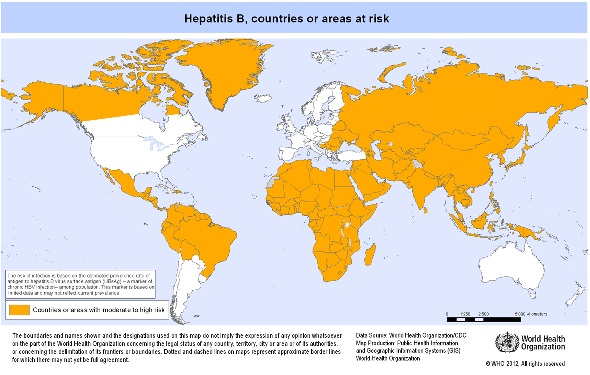
Source: World Health Organization, 2012
Risk for travellers
The risk of HBV infection for tourists and short-term travellers is usually low. However, there is an increased risk associated with certain activities, including unprotected sex with new partners, injecting drug use and participation in contact sports. Risk is typically greater in areas where there is an intermediate to high risk of HBV [2].
In 2015, 1,680 backpackers in Thailand were surveyed; 21 percent of those responding to the survey had taken part in one or more activity that may have had a risk of hepatitis B infection, including piercings, tattoos, medical or dental treatments, injecting drugs, sex with someone other than a regular partner or paid for sex. Approximately 10 percent did not always use a condom with a new partner [3].
Other travellers who may be at increased risk of exposure to hepatitis B infection include frequent, long-term, and expatriate travellers, those at occupational risk, including healthcare and humanitarian aid workers and those travelling for medical reasons or with medical conditions requiring medical treatment whilst overseas [2, 4, 5].
Transmission
HBV is spread by exposure to blood, blood products or other bodily fluids infected with HBV. It can be spread from mother to baby at birth; through a puncture wound to the skin, for example through a needlestick injury, sharing drug injecting equipment, tattooing, piercing or poor infection control during medical or dental procedures; or exposure to infected blood and body fluids for example during contact sport or through unprotected sex [2, 6].
The risk of HBV transmission through unprotected sex is high for individuals who change partners frequently, for both heterosexual relationships and men who have sex with men, and commercial sex workers [2].
In areas of high risk, hepatitis B infection is mostly passed on by perinatal transmission (infection passed from mother to baby before or after birth) or from person to person during childhood [1].
Signs and symptoms
Most people infected with HBV will have no symptoms or a mild flu-like illness. Jaundice (yellowing of the skin and eyes) occurs in about 10 percent of younger children and between 30 to 50 percent of adults [2].
Following an incubation period of 40 to 160 days, people who do experience symptoms may have jaundice, dark urine, anorexia (loss of appetite), abdominal pain, nausea, vomiting, sometimes fever and they may feel extremely tired. [1, 2]. Occasionally, severe, acute infection can cause liver failure leading to death.
Some acute HBV infections develop into a chronic (long-term) infection. The risk of developing chronic HBV infection depends on several factors including the age when first infected; As many as 90 percent of children infected in the first year of life and 30-50 percent of those infected between one and five years of age will develop long-term infection; less than 5 percent of healthy adults infected in adulthood will become chronically infected. The risk is greater for those who have a weakened immune system [2, 7].
Between 15 and 40 percent of people with long-term HBV infection develop liver damage [8]; 25 percent of people infected as children and 15 percent of people infected as adults will die prematurely [7].
Diagnosis and treatment
Hepatitis B is diagnosed by a blood test, which checks for hepatitis B antibodies or antigens (part of the virus) [9].
There is no specific treatment for acute hepatitis B, only treatment to help manage symptoms.
There are medications available for those with long-term HBV infection. They are used with the aim to slow liver damage, reduce cases of liver cancer and improve survival rates [1]. Treatment should be initiated by a specialist and follow national guidelines [9].
Preventing hepatitis B
Travellers should avoid contact with blood and bodily fluids to reduce their risk; this includes:
- Avoiding unprotected sex.
- Following universal precautions if working in healthcare or other higher risk settings.
- Avoiding tattooing, piercing and acupuncture (unless sterile equipment is used).
- Not sharing needles or other injection equipment.
- Not sharing shaving equipment.
- Awareness that medical or dental treatment abroad may be unregulated with inadequate infection control practices. See our travelling for treatment factsheet for more information.
Hepatitis B vaccination is recommended for all travellers considered to be at risk of HBV (see vaccine information below).
Travellers could consider taking a sterile medical equipment kit if travelling to resource poor areas.
Comprehensive medical insurance, covering repatriation, pre-existing medical conditions, and all planned activities, is recommended.
Travellers should be aware that using precautions against HBV will prevent other blood and body fluid-borne viruses that are not vaccine preventable, like HIV and hepatitis C.
Vaccine information
Travellers to areas with intermediate or high HBV prevalence whose activities or medical conditions put them at risk when abroad, should be offered immunisation, including those who:
- May have unprotected sex.
- May be directly exposed to blood or blood products through their occupation, such as healthcare professionals or aid workers.
- May be exposed to contaminated needles through injecting drug use.
- May be exposed to contaminated blood as a result of having medical or dental care e.g. those with pre-existing medical conditions and those travelling for medical care abroad including those intending to receive renal dialysis overseas.
- Are participating in contact sports.
- Are adopting children from a country with an intermediate or high prevalence of hepatitis B.
- Are long-stay travellers in areas of high or intermediate prevalence.
For specific vaccine recommendations see our Country Information pages.
Vaccines
The hepatitis B vaccines available in the UK are inactivated, they do not contain live organisms. They can be given as a single or combined vaccine (listed below).
Since 2017, a 6-in-1 vaccine has been given to infants as part of the UK routine childhood immunisation schedule (not listed below). For children born after 1 July 2024, a total of four doses of vaccine at the appropriate interval (ideally 8, 12, 16 weeks and 18 months of age) is recommended. For further details see the hepatitis B chapter of the 'Green Book'.
Fendrix® and HBVAXPRO 40mcg® vaccines have been developed to prevent HBV infection in patients with renal insufficiency (kidney failure), including high risk groups such as dialysis and pre-dialysis patients (these vaccines are not listed below). For further details see the hepatitis B chapter of the 'Green Book'.
The 'Green Book' and vaccine Summary of Product Characteristics (SPC) should be consulted prior to administration of any vaccine.
When using the hepatitis B monovalent vaccine (hepatitis B on its own) most risk groups should receive an accelerated schedule, given at zero, one, two and twelve months. An alternative schedule at zero, one and six months should only be used where rapid protection is not required and there is a high likelihood of compliance [2].
Health professionals should take care to review the recommended schedules of vaccines as they are age and brand specific.
Vaccine schedules for travellers
(Note: details for the infant routine vaccination schedule and those with renal failure are detailed above)
| Vaccine | Brand | Schedule(s) | Age range |
| Monovalent hepatitis B (adult) | Engerix B® Monovalent hepatitis B (20mcg/1ml) | Accelerated schedule of 4 doses: 0, 1, 2 and 12 months | 16 years and older |
| 3 doses: 0, 1 and 6 months | |||
| Very rapid schedule of 4 doses: 0, 7 and 21 days; 4th dose at 12 months | Very rapid schedule: licensed for adults 18 years and above (can consider 'off license' for 16-17 year olds) [2] | ||
| 2 doses of the adult dose: 0 and 6 months | 11 to 15 years | ||
| HBVAXPRO Adult® Monovalent hepatitis B (10mcg/1ml) | Accelerated schedule: 0, 1, 2 and 12 months | 16 years and over | |
| 3 doses: 0, 1 and 6 months | |||
| PreHevbri® Monovalent hepatitis B (10mcg/1ml): [discontinued 30 October] | 3 doses: 0, 1 and 6 months | 18 years and over | |
| HEPLISAV B® Monovalent hepatitis B (20mcg/0.5ml) | 2 doses: 0,1 months | 18 years and over | |
| Monovalent hepatitis B (paediatric) | Engerix B® Monovalent hepatitis B (10mcg/0.5ml) HBvaxPro | 3 doses: 0, 1 and 6 months | From birth to 15 years |
| Accelerated schedule: 0, 1, 2 and 12 months | |||
| HBVAXPRO Paediatric® Monovalent hepatitis B (5mcg/0.5ml) | 3 doses: 0, 1 and 6 months | From birth to 15 years | |
| Accelerated schedule: 0, 1, 2 and 12 months | |||
| Combined hepatitis A and B (adult) | Twinrix Adult® Combined hepatitis A (720ELISA units) and B (20mcg) | 3 doses: 0, 1 and 6 months | 16 years and over |
| Very rapid schedule of 4 doses: days 0, 7 and 21; 4th dose at 12 months | Licensed for adults 18 years and above (can consider 'off license' for 16-17 year olds)* | ||
| Combined hepatitis A and B (paediatric) | Ambirix® Combined hepatitis A (720 ELISA units) and B (20mcg) | 2 dose schedule given 6-12 months apart | 1 to 15 years |
| Twinrix Paediatric® Combined hepatitis A (360 ELISA units) and B (10mcg) | 3 doses: 0, 1 and 6 months | 1 to 15 years |
*Off-license use listed on the UK Health Security Agency HepA/B PGD Oct 2023.
Note that PreHevbri vaccine is no longer available in the UK. If a vaccine course has been initiated with PreHevbri, it should be completed with another adult vaccine [2].
Length of protection
The WHO has concluded that although knowledge about the duration of protection against infection and disease is still incomplete, studies demonstrate that among successfully vaccinated immunocompetent individuals, protection against acute and chronic infection persists for 20-30 years or more [10]. Therefore, there is no good evidence for recommending a booster dose of hepatitis B vaccine in routine immunisation programmes [2].
Immunocompetent adults and children who have received a complete primary course of immunisation, including children vaccinated according to the routine childhood schedule and individuals at high risk of exposure who have received a 0, 1, 6 month or 0, 1, 2, 12 months schedule, do not require a reinforcing dose of hepatitis B-containing vaccine [10].
Please be aware that the need for a booster dose following the 2-dose course of HEPLISAV B® or the 3 dose course of the now discontinued PreHevbri® has not been established.
Booster (reinforcing) doses should be considered for the following groups (see the 'Green Book' for further details):
- Healthcare and laboratory workers (including students and trainees) who have not responded to a primary course.
- People with kidney failure. A booster dose should be offered to travellers receiving haemodialysis who are intending to visit high risk countries and who have previously responded to the vaccine. This is particularly important if they are going to receive haemodialysis while away and they have not had a booster in the last 12 months.
- At the time of a significant exposure.
Testing for immunity after a course of hepatitis B vaccination is not routinely recommended, except for certain groups:
- Those at occupational risk.
- People with kidney failure.
Further guidance about testing to check for hepatitis B immunity is available in the 'Green Book' [2].
For those using a combined vaccine containing hepatitis A, the current hepatitis A guidance in the green book states that the duration of protection from a completed course of hepatitis A vaccine can be expected to be at least 25 years. Until further evidence is available, a reinforcing dose of hepatitis A vaccine is indicated at 25 years for those at ongoing risk of infection [11]. However, specific advice should be sought for individuals with altered immune responses.
Interrupted courses
It is not necessary to repeat doses if the hepatitis B course has been interrupted. Longer than recommended intervals between doses do not appear to reduce the final antibody level or efficacy. Allow the appropriate interval between the remaining doses [12].
It is good practice to continue a course of hepatitis B with the same product. However, if this is not possible, monovalent (single antigen) vaccine products may be used interchangeably, with the exception of Fendrix and HBVAXPRO40 for those with renal insufficiency. Please note that there is no published data on mixed vaccine schedules using HEPLISAV B® (and the now discontinued PreHevbri®) but they may be used if other vaccines are unavailable to avoid a delay in protection.
Contraindications
Known hypersensitivity (allergy) to any components of the vaccine, or to a previous dose. Vaccination should be delayed in those with an acute febrile illness (fever).
Undesirable effects
Adverse reactions following hepatitis B vaccine tend to be mild and short-lived. They include soreness and redness at the vaccine site. Fever, rash, malaise, and flu-like symptoms have also been reported but may not be linked to the vaccine [2]. See the individual Summary of Product Characteristics for full details.
Resources
- UK Health Security Agency: Hepatitis B information
- World Health Organization: Hepatitis
- NHS: Hepatitis B
- British Liver Trust: Hepatitis B
References
-
World Health Organization. Hepatitis B factsheet. April 2024 [Accessed 20 November 2024]
-
UK Health Security Agency. Hepatitis B. March 2025. In: Immunisation against infectious disease [Accessed 11 April 2025]
-
Frew G, McGeorge E, Grant S, de Wildt G. Hepatitis B: A cross-sectional survey of knowledge, attitudes and practices amongst backpackers in Thailand. Travel Med Infect Di. 2017, 15:57-62
-
World Health Organization. Blood safety and availability. June 2023 [Accessed 20 November 2024]
-
Harling R, Turbitt D, Millar M et al. Passage from India: an outbreak of hepatitis B linked to a patient who acquired infection from health care overseas. Public Health. 2007, 121(10):734-41 [Accessed 20 November 2024]
-
Pirozzolo JJ and LeMay DC. Blood-Borne Infections. Clinics in Sports Medicine. 2007, 26(3):425-31
-
Haber P and Schillie S. Chapter 10: Hepatitis B. Last updated 9 May 2024. In Centers for Disease Control and Prevention. Pink book. [Accessed 20 November 2024]
-
Harris A. Hepatitis B. 2024. In: Centers for Disease Control and Prevention. CDC Yellow Book [Accessed 20 November 2024]
-
NICE Clinical Knowledge Summaries. Hepatitis B. Last updated September 2024 [Accessed 20 November 2024]
-
World Health Organization. Hepatitis B vacciines: WHO position paper - July 2017 [Accessed 11 April 2025]
-
UK Health Security Agency. Hepatitis A. January 2024. In: Immunisation against infectious disease [Accessed 20 November 2024]
-
Middleman AB, Kozinetz CA, Robertson LM et al. The effect of late doses on the achievement of seroprotection and antibody titer levels with hepatitis B immunization among adolescents. Pediatrics. 2001;107(5)
-
Minor edit to reflect change to UK vaccination programme with additional dose of hepatitis B vaccine given to children at 18 months from July 2025.
-
New link to SPC added for PreHevbri vaccine, link to UKHSA green book chapter reference 2 and link for reference 10 updated.
-
PreHevbri hepatitis B vaccine now discontinued.
Explore more
Tick-borne encephalitis
Tick-borne encephalitis is a viral infection which can cause a range of symptoms from a mild and short-lived febrile illness to a severe infection of the central nervous system
Updated: 16 May 2024Chikungunya
This viral infection occurs in some tropical and subtropical regions of the world, predominantly transmitted through the bite of an infected Aedes mosquito
Updated: 12 May 2026Meningococcal disease
Meningococcal disease is a rare, but potentially devastating infection in travellers caused by the bacteria Neisseria meningitidis
Updated: 05 June 2025Yellow fever
Yellow fever is caused by a virus, which circulates between infected monkeys or humans and mosquitoes
Updated: 18 February 2026Infectious diseases
Chikungunya
This viral infection occurs in some tropical and subtropical regions of the world, predominantly transmitted through the bite of an infected Aedes mosquito
Updated: 12 May 2026Yellow fever
Yellow fever is caused by a virus, which circulates between infected monkeys or humans and mosquitoes
Updated: 18 February 2026Sex and travel: sexually transmitted infections
Advice about reducing risk of sexually transmitted infections (STIs) during travel
Updated: 09 December 2025Preparing for healthy travel
Travel Insurance
Travellers must declare medical conditions when taking out travel insurance to ensure they are suitably covered
Updated: 06 December 2021Special risk travel/traveller
Travelling with additional needs and/or disability
This factsheet provides an overview and advice on the points to consider when travelling with additional needs and/or disability
Updated: 12 September 2025Areas of humanitarian crisis
For humanitarian aid workers and those advising those travelling to areas of conflict or disaster
Updated: 07 April 2025Sickle cell disease and thalassaemia
Information on pre-travel preparation, tips to stay health abroad and links to useful resources for travellers with sickle cell disease and thalassaemia
Updated: 11 March 2024Clinic resources
Vaccines and medicines: availability, supply, shortages and use of unlicensed medicines
Information for health professionals on availability of vaccines and use of unlicensed products
Updated: 06 May 2026'Getting to Grips...' online events and video guides
Online guides and webinar sessions for health professionals interested in travel health
Updated: 05 May 2026The green book travel chapters
UK Health Security Agency Immunisation against infectious disease, the 'green book' travel chapter updates
Updated: 15 October 2024




